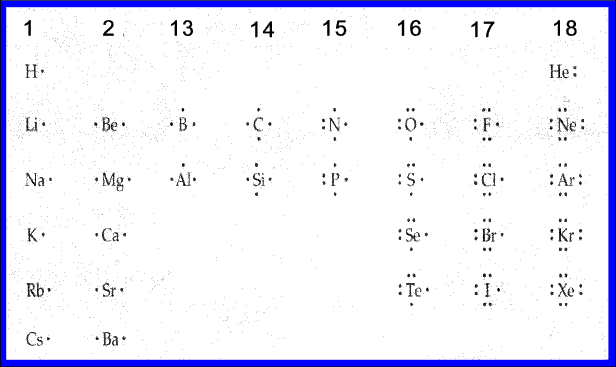
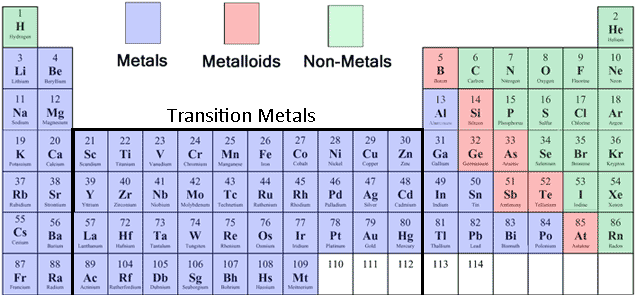
MODEL OF ATOMS, GROUPS 1-18
Molecule- a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction. (http://www.google.com/search?q=what+is+a+molecule&rlz=1CASMAE_enUS588&oq=what+is+a+mlec&aqs=chrome.1.69i57j0l5.7606j0j7&sourceid=chrome&espvd=2&es_sm=93&ie=UTF-8&safe=active&ssui=on#q=what+is+a+molecule&safe=active)
Compound- a thing that is composed of two or more separate elements; a mixture. (http://www.google.com/search?q=what+is+a+molecule&rlz=1CASMAE_enUS588&oq=what+is+a+mlec&aqs=chrome.1.69i57j0l5.7606j0j7&sourceid=chrome&espvd=2&es_sm=93&ie=UTF-8&safe=active&ssui=on#q=what+is+a+compound&safe=active)
The difference between the two is a molecule is formed together when two or more atoms join together chemically.
Compound- a thing that is composed of two or more separate elements; a mixture. (http://www.google.com/search?q=what+is+a+molecule&rlz=1CASMAE_enUS588&oq=what+is+a+mlec&aqs=chrome.1.69i57j0l5.7606j0j7&sourceid=chrome&espvd=2&es_sm=93&ie=UTF-8&safe=active&ssui=on#q=what+is+a+compound&safe=active)
The difference between the two is a molecule is formed together when two or more atoms join together chemically.
Metal- An element that readily forms positive ions (cation)
Metalloids- A chemical element that has properties in between those of metals and non-metals.
Non-metals- A chemical element which mostly lacks metallic attributes.
Transition metals- The 38 elements in groups 3-12 of the periodic table.
HOW TO DETERMINE THE NUMBER OF PROTONS, ELECTRONS, AND NEUTRONS IN AN ELEMENT
https://www.youtube.com/watch?v=g8zADkO5trc
How to determine the number of valence electrons on any given element: https://www.youtube.com/watch?feature=player_detailpage&v=x1gdfkvkPTk
-Elements are in different groups because of their different properties. They are divided by types of metals and gases.
-Mass is the amount of matter an object has. You usually would use a triple balance beam to measure mass. To measure an objects volume you have to use the volume formula for the shape. To calculate an objects density, you have to divide the mass over the volume.
-The difference between a physical and chemical property is A physical property is an aspect of matter that can be observed or measured without changing it. A chemical property may only be observed by changing the chemical identity of a substance.
-Phase changes are physical properties because a phase change is a change from one state (solid or liquid or gas) to another without a change in chemical composition.
-Evaporation is a physical property because water can be in three physical states.
Examples of physical and chemical changes:
1. Physical- Melting an ice cube
2. Physical- Crushing a can
3. Chemical- Burning wood
4. Chemical- Digesting food
Here's an experiment showing both physical and chemical changes. http://www.youtube.com/watch?feature=player_detailpage&v=kFrn9I-ijr4
To balance an equation, you must add matter to both sides until there are equal numbers of each type of atom on both sides of the equation. Here's a video. https://www.youtube.com/watch?v=ZxQp80Fvhjo&feature=player_embedded
www.chemistry.com
www.youtube.com
Metalloids- A chemical element that has properties in between those of metals and non-metals.
Non-metals- A chemical element which mostly lacks metallic attributes.
Transition metals- The 38 elements in groups 3-12 of the periodic table.
HOW TO DETERMINE THE NUMBER OF PROTONS, ELECTRONS, AND NEUTRONS IN AN ELEMENT
https://www.youtube.com/watch?v=g8zADkO5trc
How to determine the number of valence electrons on any given element: https://www.youtube.com/watch?feature=player_detailpage&v=x1gdfkvkPTk
-Elements are in different groups because of their different properties. They are divided by types of metals and gases.
-Mass is the amount of matter an object has. You usually would use a triple balance beam to measure mass. To measure an objects volume you have to use the volume formula for the shape. To calculate an objects density, you have to divide the mass over the volume.
-The difference between a physical and chemical property is A physical property is an aspect of matter that can be observed or measured without changing it. A chemical property may only be observed by changing the chemical identity of a substance.
-Phase changes are physical properties because a phase change is a change from one state (solid or liquid or gas) to another without a change in chemical composition.
-Evaporation is a physical property because water can be in three physical states.
Examples of physical and chemical changes:
1. Physical- Melting an ice cube
2. Physical- Crushing a can
3. Chemical- Burning wood
4. Chemical- Digesting food
Here's an experiment showing both physical and chemical changes. http://www.youtube.com/watch?feature=player_detailpage&v=kFrn9I-ijr4
To balance an equation, you must add matter to both sides until there are equal numbers of each type of atom on both sides of the equation. Here's a video. https://www.youtube.com/watch?v=ZxQp80Fvhjo&feature=player_embedded
www.chemistry.com
www.youtube.com